Investigation: What Factors Affect Photosynthesis
Background and PreLab
Photosynthesis fuels ecosystems and replenishes the Earth's atmosphere with oxygen. Like all enzyme-driven reactions, the rate of photosynthesis can be measured by either the disappearance of substrate, or the accumulation of products. The equation for photosynthesis is:
6CO2 + 6H2O ------light--------> C6H12O6 + 6O2 + H20
The rate of photosynthesis can be measured by:
1) measuring O2 production 2) measuring CO2 consumption
Leaf Structure and Function
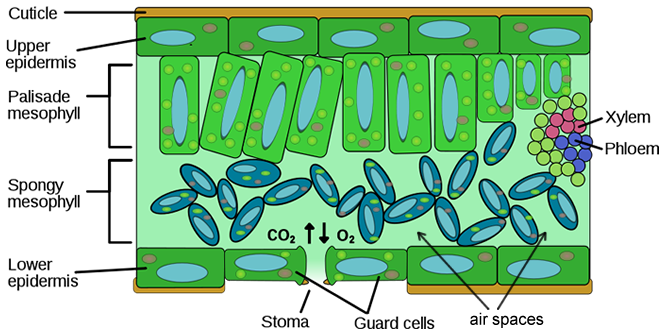
In this investigation, you will use a system that measures the accumulation of oxygen in the leaf. Consider the anatomy of the leaf as shown below.
The leaf is composed of layers of cells. The spongy mesophyll layer is normally infused with gases, oxygen and carbon dioxide. Leaves (or disks cut from leaves) will normally float in water because of these gases. If you draw the gases out from the spaces, then the leaves will sink because they become more dense than water. If this leaf disk is placed in a solution with an alternate source of carbon dioxide in the form of bicarbonate ions, then photosynthesis can occur in a sunken leaf disk. As photosynthesis proceeds, oxygen accumulates in the air spaces of the spongy mesophyll and the leaf becomes buoyant and floats. Oxygen and carbon dioxide are exchanged through openings in the leaf called stoma.
While this is going on, the leaf is also carrying out cellular respiration. This respiration will consume the oxygen that has accumulated and possibly cause the plant disks to sink. The measurement tool that can be used to observe these counteracting processes is the floating (or sinking) of the plant disks. In other words, the buoyancy of the leaf disks is actually an indirect measurement of the net rate of photosynthesis occurring in the leaf tissue.
Learning Objectives:
1) To design and conduct an experiment to explore factors that affect photosynthesis.
2) To connect and apply concepts, including the relationship between cell structure and function, strategies for capture and stores of energy, and the diffusion of gases across membranes.
Experimental Question: What factors affect the rate of photosynthesis?
PreLab Questions - these should be completed BEFORE the scheduled lab
1. How can the rate of photosynthesis be measured?
2. Where in the cells of the leaf do you find air spaces?
What is the function of the stomata?
3. What will happen if you remove the air from these spaces?
4. How will air return to these spaces?
5. Instead of carbon dioxide, what will be used as the reactant in this lab?
6. List any factors that you think may affect the rate of photosynthesis. Consider environmental factors that you could manipulate during the lab.
Part 1: Basic Procedure for Measuring the Rate of Photosynthesis
Materials: baking soda, liquid soap, plastic syringes, leaves (spinach or ivy), hole punch, cups or beakers, timer, light source
1. Collect leaf disks by punching holes in the leaf (try to get them between the veins), you will need 20 leaf circles.
2. Make a solution of sodium bicarbonate by mixing 300 ml of water to a a pinch of baking soda (about 100 ml to 1g)
3.
Make a diluted solution of liquid detergent in a small beaker by adding 2 drops of dish soap to 100 ml of water. Do not make suds!
4.
Add one drop of this dilute soap solution to your 300 ml bicarbonate solution. Swirl gently to avoid making suds.
5.
Place 10 leaf disks into the syringe and pull in a small volume of the bicarbonate and soap solution. Replace the plunger and push out most of the air, but do not crush your leaves.
6. Create a vacuum by covering the tip of the syringe with your finger. Draw back on the plunger.
7. Release the vacuum so that the solution will enter the disks. It may take a few times to get the disks to sink. You may need to gently tap the syringe to dislodge discs from the sides.
8. Once they sink, you can put them back into the sodium bicarbonate solution and expose the disks to light. Start a timer and record how many of the disks are floating at 1 minute intervals. (See data table.) While you are making observations, you can set up the control group.
Troubleshooting: Gently swirl the solution to dislodge disks which may become stuck to the bottom. If no disks float within 5 minutes, add a couple more drops form your soap solution and start the time over again. Place you beaker as close to the light as possible.
9.Control Group: Repeat your set-up from above, but this time do not place baking soda in the beaker. Place another set of sunken disks into this solution and record data on the table.
10. Both the experimental group and the control should run until all the discs are floating.
Data Table
| Time (min) | # of floating disks (bicarbonate, water, + soap) |
# of floating disks (control) ( only water + soap) |
| 1 | ||
| 2 | ||
| 3 | ||
| 4 | ||
| 5 | ||
| 6 | ||
| 7 | ||
| 8 | ||
| 9 | ||
| 10 | ||
| 11 | ||
| 12 | ||
| 13 | ||
| 14 | ||
| 15 |
Analyzing Data
To make comparisons between experiments, a standard point of reference is needed. Repeated testing of this procedure has shown that the point at which 50% of the disks are floating (the median or ET50) is a reliable and repeatable point of reference. In this case, the disks floating are counted at the end of each time interval. The median is chosen over the mean as the summary statistic.
The median will generally provide a better estimate of the central tendency of the data because, on occasion, a disk fails to rise or takes a very long time to do so. A term coined by G. L Steucek and R. J Hill (1985) for this relationship is ET50, the estimated time for 50% of the disks to rise. That is, rate is a change in a variable over time. The time required for 50% of the leaf disks to float is represented as Effective Time = ET50.
Graph your data for the experimental group. Determine the ET50 for your leaf disks and determine the ET50 for your data.
What is the relationship between sodium bicarbonate and photosynthesis rate? This is your CLAIM.
Provide evidence that supports this claim; summarize data by referencing ET50.
Provide reasoning that linke the evidence and the claim and explains why this relationship exists.
Part 2: Design and Conduct Your Own Investigation
Now that you have mastered the floating disk technique, you will design an experiment to test another variable that may affect the rate of photosynthesis. You will collect data, analyze data and present your findings in the form of a LAB REPORT. As you conduct your investigation, you may want to take photos to include in your report. Choose from the list of variables below to investigate. (If you have another variable that you would like to try, check with your instructor first.)
light intensity or distance from the light | amount of sodium bicarbonate
water temperature | size of leaf disks or shape of leaf disks | color of light
1. Describe your experiment. You may use sketches.
2. Compile your results into a table and graph the results showing the ET 50
3. Summarize the results of the experiment using the CER format.
Other Resources on Photosynthesis
Inquiry Lab with Vernier Probes
Separation of Plant Pigments with Chromatography
Light Dependent Reaction and Calvin Cycle